-
Address:
17888 67th Court North
Loxahatchee, FL
-
Mail us:
contact@wrightacademia.org
- submit manuscript
Table of content
Research Article |
Open Access |
Volume 1 | Issue 1 |
Cardiac Biomarkers in Acute Stroke
Fiori Patrizia1, Capaldo Guglielmo1, Corbo Antonio1, Corbo Giulia1, Di Gregorio Maria1, Iorillo Luigi1, Pelosi Chiara2, Savino Patrizia2, Botticella Filomena3, Dragonetti Carmela3, Manganiello Gianvito3, Morella Alessandro3, Pellecchia Vincenzo3, Gennaro Bellizzi3, Alberico Marielisa4, Benigni Giovanni4, De Caro Monica4, Guerriero Barbara4, Pace Erminio4, Raffa Marianna4, Ferrara Maurizio4, Mazza Emerico5, Tammaro C.A.6, Giannetti L.M.7 and Monaco Antonio1
1Neurological Unit, S. Ottone Frangipane Hospital, Ariano Irpino (AV), ASL AV, University of Naples, Italy
2Internal Medicine, S. Ottone Frangipane Hospital, Ariano Irpino (AV), ASL AV, University of Naples, Italy
3Cardiological Unit, S. Ottone Frangipane Hospital, Ariano Irpino (AV), ASL AV, University of Naples, Italy
4Intensive Care, S. Ottone Frangipane Hospital, Ariano Irpino (AV), ASL AV, University of Naples, Italy
5Radiology, S. Ottone Frangipane Hospital, Ariano Irpino (AV), ASL AV, University of Naples, Italy
6Laboratory, S. Ottone Frangipane Hospital, Ariano Irpino (AV), ASL AV, University of Naples, Italy
7Infantile Neuropsychiatry, S. Ottone Frangipane Hospital, Ariano Irpino (AV), ASL AV, University of Naples, Italy
*Corresponding author: Dr. Fiori Patrizia, Neurological Unit, S. Ottone Frangipane Hospital, Ariano irpino (AV), ASL AV, University of Naples, Italy Tel: 39-825-877602, Fax: 39-825-828409.
Citation: Patrizia F, Guglielmo C, Antonio C, Giulia C, Maria DG, et al. (2019) Cardiac Biomarkers in Acute Stroke. Academia J Stroke. 1:002.
Copyright © Patrizia F, et al.
Received: |
Accepted: |
Published: |
Background
Serum cardiac biomarkers are increased in cerebrovascular diseases. The aim of our study was toassess their levels according to the severity of cardiological and neurological conditions.
Methods and results
We recruited 552 acute ischaemic stroke (AS), 290 chronic cerebrovascular (CCVD) and 111 other neuropsychiatric diseases (OND) patients. Blood withdrawals were performed within 12-24 hours. Serial assessments were repeated at day 3 and 7.
At admission, high sensitive T Troponin (hs T Tro) and N-Terminal Pro-Brain Natriuretic Peptide II (NT-pro- BNP) levels were increased in AS and CCVD compared to OND (inter-group variability), especially in patients in class III/IV, C/D New York Heart Association and American Cardiology Association scales. Signs of acute myocardial infarction were observed in a minority of patients. No significant fluctuations were found within each group of patients at repeated measurement in one-week time (intra-group unvariability) at ANOVA repeated measures. However, delta criterion allowed identifying malignant cerebrovascular profile. ASPECTS scores were tendentially lower in patients in class III/IV New York Heart Association, C/D American Cardiological Association scales with stable or increased levels of NT-pro-BNP compared to those with 100% relative percentage decrease of such biomarker at day VII. Better clinical course and prognosis were observed in AS patients with at least 50% relative percentage decrease of NT-pro-BNP at day 7.
Conclusions
Although serum cardiac biomarkers may reflect more the chronicity rather than the acuity of ischaemic sufferance, delta criterion helps in differential diagnosis between acute versus chronic condition. NT-pro-BNP may be an early warning marker of either an adaptive or maladaptive response, with volume and pressure overload preceding hs T Tro rise and irreversible systemic, ischaemic damage. Moreover, it is reliable for assessing the severity of clinical conditions and the prognosis 279.
Introduction
In clinical practice, it is pivotal to identify useful parameters for prompt diagnosis, continuous monitoring, correct treatment, definite prognosis, safe discharge, scheduled follow up. Two cardiac biomarkers, high sensitive T Troponin (hs T Tro) and Brain Natriuretic Peptide (BNP), are commonly assessed in emergency department and for monitoring clinical conditions during hospitalization.
T Tro is a structural myocardial molecule. In myocardial necrosis, the entity of its release is a marker of the severity of acute coronary syndrome [1]. Its level predicts all-cause mortality [2-4], correlates with stroke severity, disability, increased brief and long-term mortality [5-11]. However, the dynamic relative and absolute changes are prognostically more significant than stable elevation in chronic conditions. National Academy of Clinical Biochemistry suggests using the delta criterion (changes greater than 20%) in sensitive assay when the value is above 99th percentile [12,13].
BNP is synthesized and released by cardiomyocytes in response to cardiac haemodinamic stress with increased transmural wall tension (heart volume and pressure overload). Its level tends to be higher in female gender and increases by aging [14,15]. It is considered a marker of cardiac impairment, as in left ventricular dysfunction and cardiac heart failure [16-19]. N-Terminal Pro-Brain Natriuretic Peptide II (NT-pro-BNP) results from cleavage of the precursor molecule, namely pro-BNP. It is considered the best diagnostic and prognostic parameter in cardiac heart failure [20]. It is a strong predictor for coronary artery disease severity and correlates with left ventricular ejection fraction [21]. Low in-hospital reduction and high discharge level are independent markers for death or readmission after decompensated chronic heart failure [22,23]. Intensive care survivors had significantly lower NT-pro-BNP values than non-survivors [24]. This parameter predicts risk of stroke [25-29], its clinical and radiological severity, recurrence and poor prognosis [30-37], cardioembolic genesis [38].
Both cardiovascular biomarkers are associated with increased risk of all-cause mortality [39-41].
The aim of our study was to evaluate hs T Tro and NT-pro-BNP levels in patients affected with acute stroke and/or chronic cerebrovascular conditions 313.
Materials and Methods
Nine-hundred-fifty-three patients, 552 (57.9%) of which affected with acute ischaemic stroke (AS) (age 78.6 sd 11.5), 290 (30.4%) with chronic cerebrovascular (CCVD) (age 77.3 sd 8.7) and 111 (11.6%) with other neuropsychiatric diseases (OND), as psychoses, epilepsies, minor traumas, (age 49.2 sd 15.4), were recruited in our Neurological Unit/Spoke Unit (Table 1). Blood withdrawals were performed within 12-24 hours after admission. Serial assessments were repeated at day 3 and 7. Results were analyzed in subgroups of patients according to the severity of heart and neurological dysfunction, evaluated by the CHAD2DS2VAsc, HAS BLED, New York Heart Association (NYHA), American Cardiology Association (ACA) scales, Simplified Pulmonary Embolism Severity Index (SPESI), Pulmonary Embolism Severity Index (PESI), Apache score, Glasgow Coma (GCS)/Outcome (GOS) scales, Hachinski and Modified Rankin Scale (MRS) scales. ECG, transthoracic and/or transesophageal echocardiography were performed within one-week time. We focalized our attention on the following echocardiographic parameters: ejection fraction (EF, normal range 60-80%), pulmonary arterial pressure (PAP, normal < 25 mmHg), atrial dimension (AD, normal range 3.3 sd 0. 5 cm). All patients underwent Computerized Tomography and/or Magnetic Resonance Imaging at admission, after 24 hours and, if necessary, repeated.
Cardiac biomarkers were detected according to standard methods. Serum hs T Tro and NT-pro-BNP II levels were measured by electrochemiluminescence immunoassay with biotinylated monoclonal anti-mouse-T Tro and anti-mouse-NT-pro-BNP-specific antibodies labeled with ruthenium, which formed a sandwich complex. After addition of streptavidin-coated microparticles, it bound to solid phase. The microparticles were aspired and magnetically captured by an electrode, while unbound particles were removed. Voltage induced chemoluminiscence was revealed by Cobas analyzer (Elecsys and Cobas, Roche). The cut off value to rule out myocardial infarction was < 15 pg/ml for hs T Tro. Normal range value of NT-pro-BNP were 0-125 pg/ml.
Statistical analysis was performed by unpaired T test, ANOVA repeated measures, for standard description of baseline characteristics and differences among the studied groups, by Pearson correlation test and regression analysis, for identification of association among examined parameters. Moreover, delta criterion was applied, considering differences of at least 20% for hs T Tro and 50% for NT-pro-BNP 345.
Results
Increased hs T Tro and NT-pro-BNP were detected in AS (hs T Tro: 59.6 sd 356.3 pg/ml, p 0.02; NT- pro-BNP: 3103.6 sd 5772.6 pg/ml, p 0.01) and CCVD (hs T Tro: 28.1 sd 33.6 pg/ml, p 0.03; NT-pro-BNP: 1031.6 sd 2462.2 pg/ml, p 0.03) compared to OND (hs T Tro: 6.8 sd 4.5 pg/ml; NT-pro-BNP: 124.7 sd 245.5 pg/ml) at T test (inter-group variability) (Figure 1). Levels above normal values of hs T Tro were present in 373 (67.5%) AS, 169 (58.2%) CCVD, 7 (6.3%) OND, of NT-pro-BNP in 477 (86.4%) AS, 220 (75.8%) CCVD, 32 (28.8%) OND.
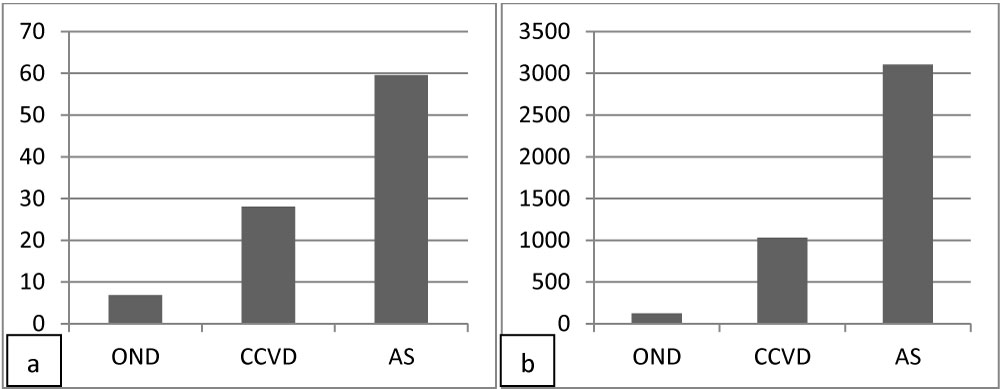
Figure 1: Levels of hs T Tro (a) and NT-pro-BNP (b) in OND, CCVD and AS patients at admission.
Three-hundred-fifty-three (63.9%) AS, 106 (36.6%) CCVD and two (1.8%) OND were classified in class III/IV NYHA, C/D ACA (Table 1). Significant differences were found between patients in class I/II, A/B compared to class III/IV, C/D NYHA and ACA scales in AS (hs T Tro 18.9 sd 27.6 vs. 79.7 sd 380.7 pg/ml, p 0.001; NT-pro-BNP 241.8 sd 238.5 vs. 7202.1 sd 4821.5 pg/ml, p 0.01) (Figure 2).
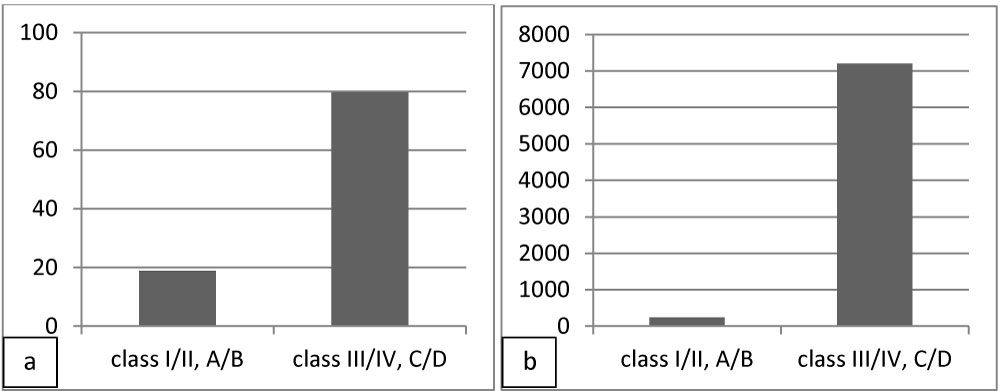
Figure 2: Levels of hs T Tro (a) and NT-pro-BNP (b) in class I/II NYHA, A/B ACA and class III/IV NYHA, C/D ACA AS patients at admission.
Moreover, the subgroup of AS with CCVD had higher levels compared to AS without CCVD (hs T Tro: 50.1 sd 117.9 vs.17.6 sd 27.7 pg/ml, p 0.01; NT-pro-BNP 3609.5 sd 6121.7 vs. 1092.1 sd 3449.8 pg/ml, p 0.03) (Figure 3).
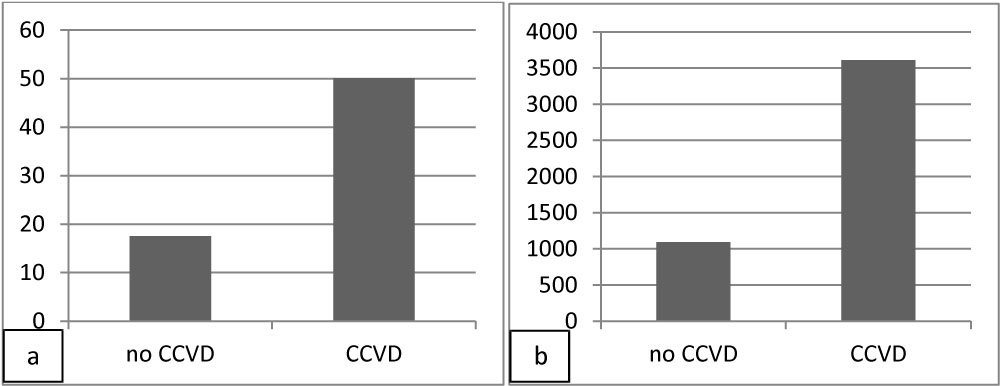
Figure 3: Levels of hs T Tro (a) and NT-pro-BNP (b) in AS patients without or with CCVD at admission.
Signs of acute myocardial necrosis were observed in a minority of patients, 3 (2.7%) OND, 3 (1%) CCVD, 20 (3.6%) AS at electrocardiograms, 5 (4.5%) OND, 14 (4.8%) CCVD, 40 (7.2%) AS at echocardiograms.
Supraventricular extrasystolia was present in 30 (27%) OND, 61 (21%) CCVD, 94 (17%) AS, ventricular extrasystolia in 20 (18%) OND, 61 (21%) CCVD, 94 (17%) AS, blocks of branches in 12 (10.8%) OND, 61 (21%) CCVD, 110 (19.9%) AS, atrioventricular blocks in 2 (1.8%) OND, 14 (4.8%) CCVD, 22 (4%) AS, atrial fibrillation in 38 (13.1%) CCVD, 166 (30%) AS, minor valvular dysfunctions in 28 (25.2%) OND, 159 (54.8%) CCVD, 267 (48.4%) AS, moderate-severe valvular cardiopathy in 44 (15.2%) CCVD, 144 (26.1%) AS (Table 1). The reliability of cardiac biomarkers concerning myocardial infarction is shown in Table 2. Significant correlation was observed between levels of NT-pro-BNP and severity of cardiac dysfunction evaluated by NYHA and ACA scales, the severity of which were scored in the range of 1 to 4 (r 0.50).
Table 2: The reliability of cardiac biomarkers concerning myocardial infarction. View Table 2
Serial assessment did not show significant fluctuations within each groups of patients at day 3 and 7 compared to admission (intra-group unvariability) (Figure 4) and at different time lag in bounce backs (preliminary data, not shown) at ANOVA repeated measures.
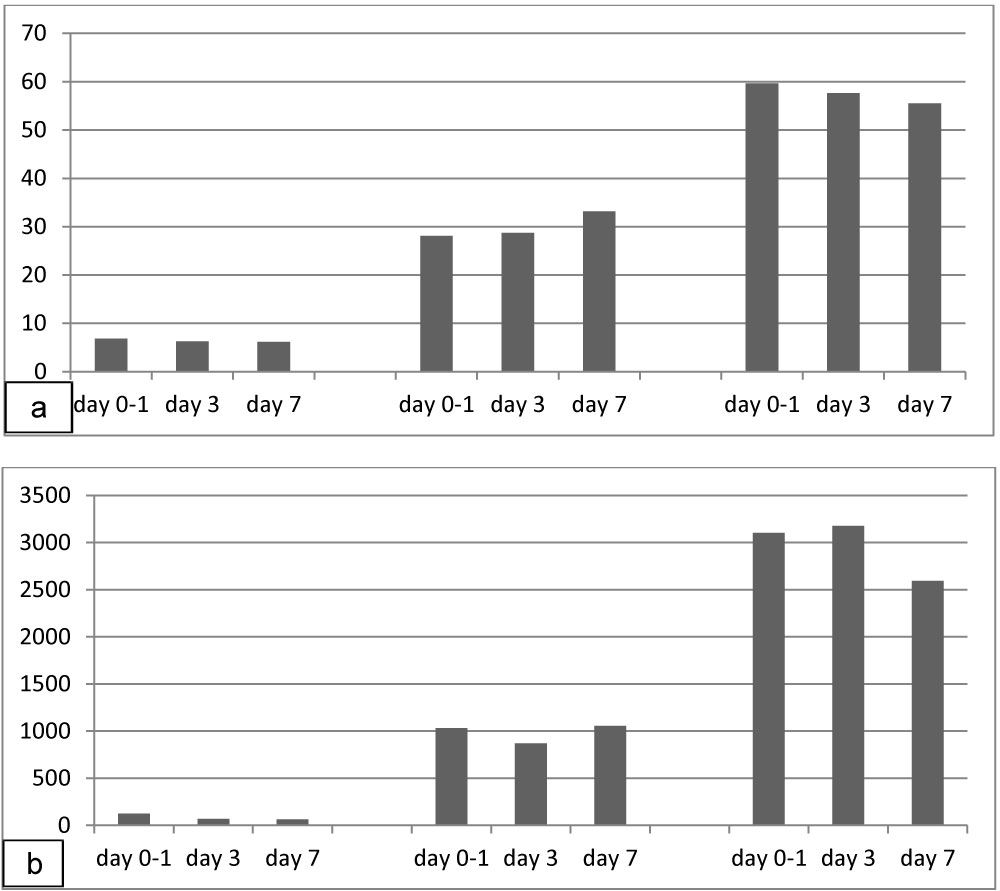
Figure 4: Serial assessment of hs T Tro (a) and NT-pro-BNP (b) at day 0-1, 3 and 7 in patients affected with OND (left columns), CCVD (columns at center), AS (right columns).
Overall, according to the delta criterion, significant relative decrease > 20% of hs T Tro was found in CCVD (- 31.16%) (Figure 5a). Significant relative percentage decreases > 50% of NT-pro-BNP were observed in OND (-124.2%), in CCVD (-58.04%) and in AS (-91.17%) (Figure 5b). Relative percentage decreases > 20% of hs T Tro were present in 28 (25.2%) OND, 85 (29.3%) CCVD, 186 (33.7%) AS. Relative percentage decreases > 50% of NT-PBNP were detected in 58 (52.3%) OND, 103 (35.5%) CCVD, 235 (42.6%) AS. No significant relative percentage changes were revealed in 65 (58.6%) OND, 157 (54.1%) CCVD, 243 (44%) AS concerning hs T Tro, 44 (39.6%) OND, 156 (53.8%) CCVD, 248 (44.9%) AS concerning NT-pro-BNP.
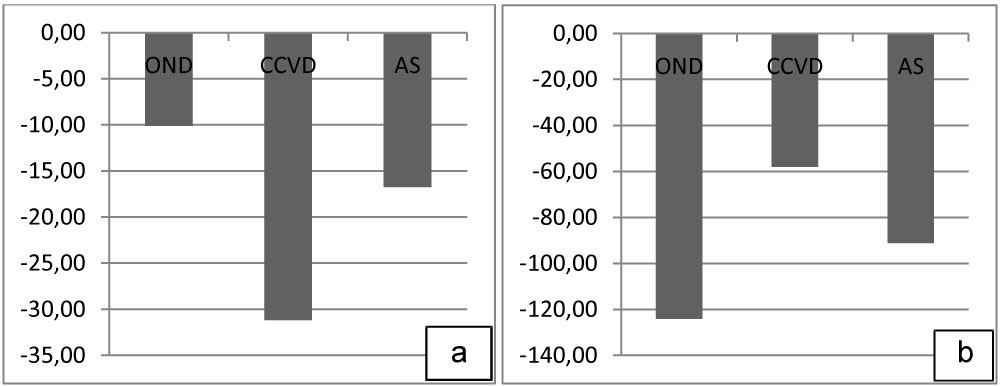
Figure 5: Relative percentage decreases hs T Tro (a) and NT-pro-BNP (b) in OND, CCVD and AS patients at day 7.
Significant differences were present between AS with relative decrease of at least 50% in NT-pro-BNP levels compared with AS without significant changing of such marker regarding GCS (12.6 sd 2.7 vs. 11.9 sd 2.9, p 0.02), pre-stroke MRS (1 sd 1.46 vs. 1.34 sd 1.56, p 0.03), Hachinski scale (9.9 sd 2 vs. 10.4 sd 2.1, p 0.01), HAS (4.2 sd 1 vs. 4.4 sd 1, p 0.02) at admission, GCS (13.6 sd 2.1 vs. 12.8 sd 2.6, p 0.0004), GOS (3.8 sd 0.8 vs. 3.5 sd 0.8, p 0.0001), MRS (2.6 sd 1.7 vs. 3.1 sd 1.6, p 0.0006) at day 7 (Table 3). The highest levels of hs T Tro were detected in class III/IV, C/D patients with at least 50% relative increase compared to those with at least 50% relative decrease of NT-Pro-BNP at day VII. These patients had the lowest GCS at admission, as well as the lowest GCS and highest MRS at day VII.
Table 3: Significant differences were present between AS with relative decrease of at least 50% in NT-pro-BNP levels compared with AS without significant changes. View Table 3
In class 0/I OND, a significant reduction of NT-pro-BNP levels was already detected at day 3 (38.7 sd 34.6 vs. 77.2 sd 71.3, p 0.0001), especially in females, compared to males (females 45.7 sd 30.4 vs. 103.3 sd 74.2, p 0.0002; males 33.1 sd 37.1 vs. 55.2 sd 61.6, p 0.06). This finding was confirmed at day 7 (40.3 sd 54.3 vs. 77.2 sd 71.3, p 0.01; females 43.3 sd 28.3 vs. 103,3 sd 74.2, p 0.007; males 37.2 sd 73 vs. 55.2 sd 61.6, p 0.08) (Figure 6). Any stressful event, diagnostic procedures included, were responsible of fluctuations of such marker.
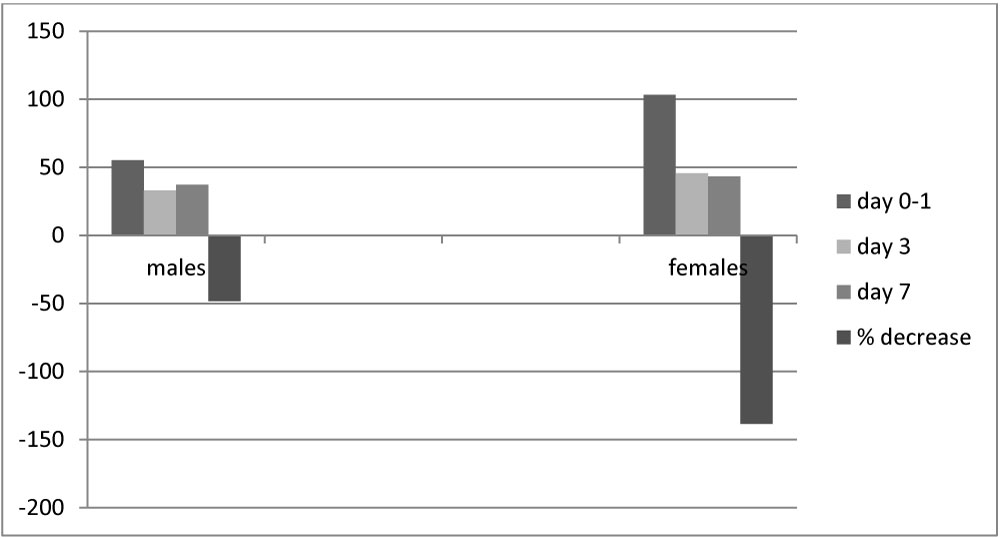
Figure 6: Serial assessment of NT-pro-BNP at day 0-1, 3 and 7 in male (left) and female (right) patients affected with OND and respective relative percentage decreases at day 7.
Significant differences were observed in AD between OND, CCVD and AS (OND 3.5 sd 0.6 cm vs. CCVD 4.2 sd 0.6 cm, p 0.03; OND 3.5 sd 0.6 cm vs. AS 4.8 sd 4.3 cm, p 0.01) (Figure 7). The most significant correlations were present between hs T Tro and EF (r -0.22), AD (r 0.28), PAP (r 0.23), between NT-pro-BNP and EF (r -0.24), AD (r 0.25), PAP (r 0.48) in CCVD, between NT-pro-BNP and EF (r -0.34), PAP (r 0.47) in AS.
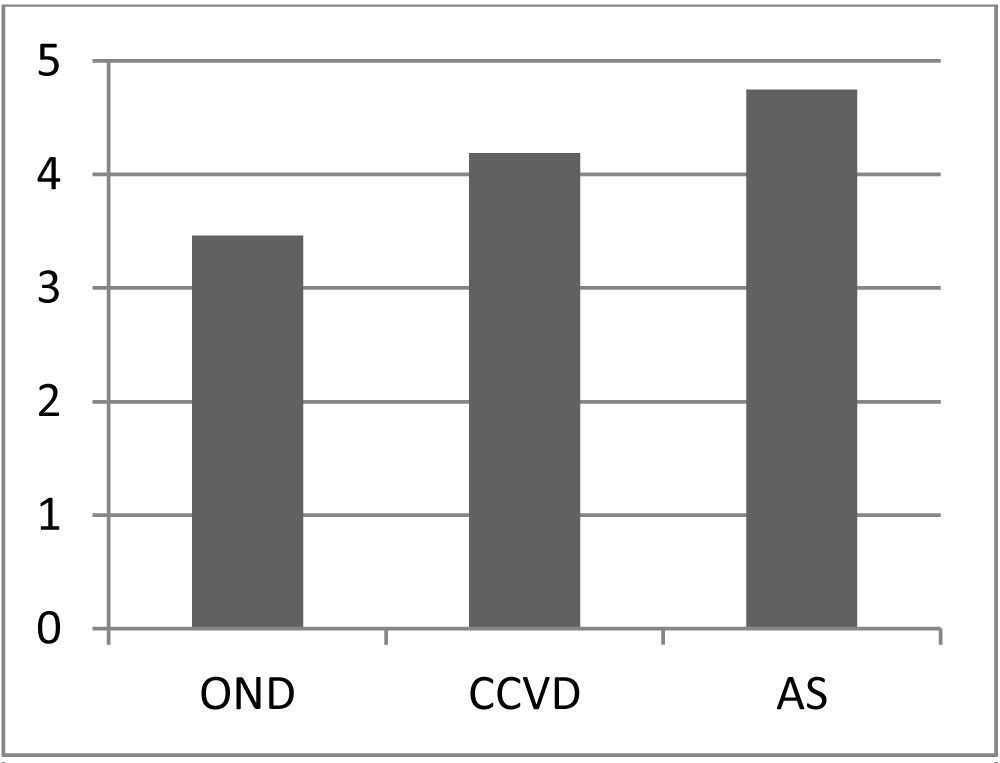
Figure 7: Atrial diameters in OND, CCVD and AS patients.
Lacunes were present in all CCVD and in 61 (11.1%) AS. Signs of previous ischaemic strokes were evident in 38 (13.1%) CCVD and 83 (15%) AS. Acute infarcts were in the territory of anterior circulation in 320 (58%) AS, in vertebro-basilar areas in 110 (19.9%) AS. Haemorrhagic infarction occurred in 22 (4%) AS. Other 122 (22.1%) AS were considered lacunar. No significant differences of cardiac biomarkers were related to the region of the cerebral infarction (Internal Carotid Artery, Middle Cerebral Artery, Anterior Cerebral Artery AS: hs T Tro 73.4 sd 430.2 pg/ml, NT-pro-BNP 3898.6 sd 6648.4 pg/ml; Vertebro-Basilar AS: hs T Tro 60 sd 153.5 pg/ml; NT-pro-BNP: 3607.9 sd 6143.7 pg/ml, p ns). This was confirmed in subgroups' analysis among cardiac biomarkers in fronto-parietal (hs T Tro 24.93 sd 19.37 pg/ml; NT-pro-BNP 1717.1 sd 1996.4 pg/ml), temporo-insular (hs T Tro 23.9 sd 15.1 pg/ml; NT-pro-BNP 2581 sd 4166.1 pg/ml), capsulo-nuclear (hs T Tro 27.73 sd 27.27 pg/ml; NT-pro-BNP 2022.4 sd 2435.5 pg/ml), occipital (hs T Tro 41.68 sd 42.57 pg/ml; NT-pro-BNP 2398.8 sd 2198.4 pg/ml), cerebellar-brainstem (hs T Tro 30.6 sd 26.4 pg/ml; NT-pro-BNP 2080.8 sd 2771.9 pg/ml) AS (Figure 8). Their levels as the extension of cerebral ischaemia were related to the overall severity of neurocardiological dysfunction, responsible of diffuse hypoxic encephalopathy in severe cases. In patients with early neuroradiological signs of cerebral ischaemia at admission, ASPECTS scores were tendentially lower in patients with stable or increased levels of NT-pro-BNP compared to those with at least 100% relative decrease of NT-pro-BNP at day VII (class I/II NYHA, A/B ACA patients 5.33 sd 1.56 vs. 5.83 sd 1.6, p ns; class III/IV NYHA, C/D ACA 4.25 sd 1.71 vs. 5.03 sd 1.66, p 0.07) (Figure 9). In the latter, significant correlations were found between ASPECTS, GCS (r 0.47) at admission, GCS (r 0.30), MRS (r -0.20), hs T Tro (-0.58) at day VII 1192.
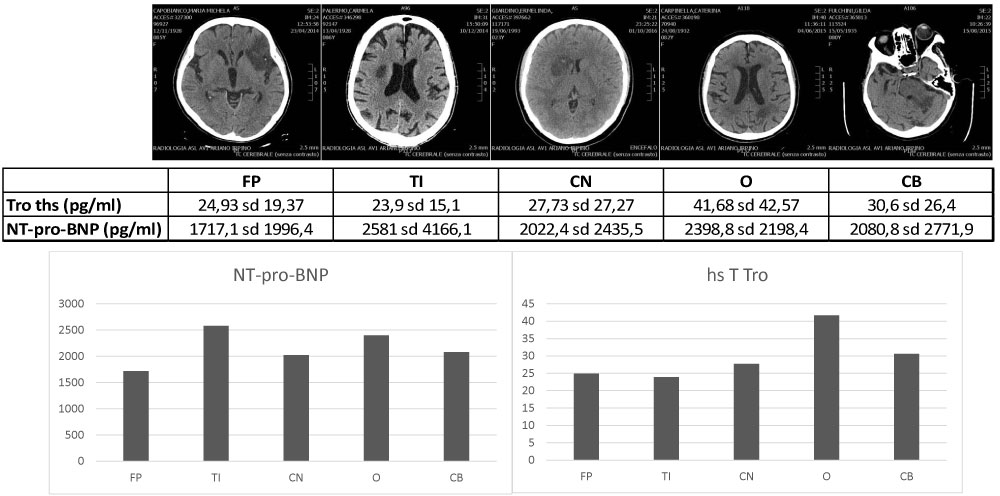
Figure 8: NT-pro-BNP and hs T Tro levels in fronto-parietal (FP), temporo-insular (TI), capsule-nuclear (CN), occipital (O), cerebellar-brainstem (CB) AS.
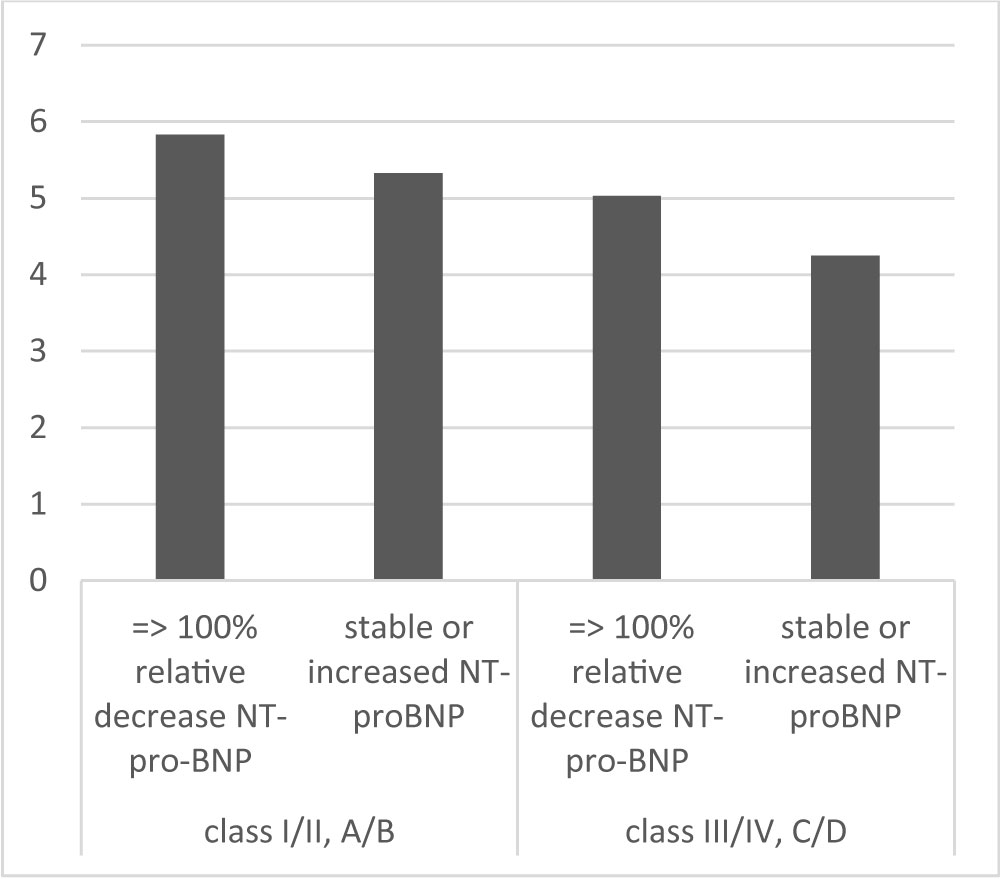
Figure 9: ASPECTS scores and relative percentage changes of NT-pro-BNP at day VII in AS patients.
Discussion
Cardiac markers are increased in cerebrovascular conditions, particularly in AS in class III/IV, C/D NYHA and ACA scales and in the contest of chronic cerebrovascular disease (CCVD), compared to other neurological diseases (OND) (inter-group variability). No significant fluctuations were found within each group of patients at repeated measurements in one-week time (intra-group unvariability) and at different time lag in bounce backs (preliminary data). However, significant absolute and relative changes of such parameters are pivotal for early detection of sudden acuity or worsening of chronicity. Moreover, their decreases are associated with better outcomes while their increases point out a malignant prognosis.
NYHA scale allows a quick assessment of the severity of cardiological conditions. Together with cardiac biomarkers speed up diagnostic iter for appropriate emergency treatments. According to cardiologic criteria, since cardiac biomarkers reveal myocardial injury, independently on the etiopathogenetic cause, their elevation is not sufficient for a definite diagnosis of acute coronary syndrome. At least one of the following clinical criteria has to be present: symptoms of ischemia, electrocardiogram changes (new ST-T changes or Left Bundle Branch Block, new Q waves), imaging evidence, such as a new regional wall motion abnormality, new loss of viable myocardium on the echocardiogram or intracoronary thrombus at coronaroangiography [42]. We found significant ECG/echocardiographic acute ischaemic signs in a minority of our patients. Therefore, we highlight the value of cardiac biomarkers for risk evaluation and for monitoring clinical conditions. Moreover, their levels predict the severity at ACA scale.
However, in real-world practice, comorbidities are frequent, reduce penumbra and collaterals, account for increased levels of both T Tro and NT-pro-BNP. In Dallas Heart Study, up to 40% of patients have troponin levels in the range of myocardial infarction [43]. Association with age, male gender, trauma, burns, inflammation, hypertension, diabetes, heart failure, cerebrovascular accidents, pulmonary emboli, renal impairment is reported. More than 90% of patients with stable coronary artery disease, without heart failure, have levels of T Tro within the measuring range [44]. Nonetheless, the utility of delta criterion to distinguish acute from chronic conditions is undeniable. In this regard, we observe that relative percentage decreases ought to be considered together with absolute levels and changes, especially in critical conditions (e.g. the relative percentage decrease has a different meaning if the absolute value is in the range of thousand or ten picograms). Prognostic sensibility of NT-pro-BNP seems to be even higher than that of T Tro [45,46]. Among cardiac biomarkers, NT-pro-BNP has the features of an ideal biomarker, since it is released quickly for early diagnosis, it is persistent for a reasonable amount of time to allow a diagnostic window (half-life 90-120 minutes) and can be quantified accurately and economically, reflecting changes in both the patient's status and prognosis [47]. NT-pro-BNP levels resulted to be a stronger prognostic marker than ECG and its dosage has been proposed as first test for cardiovascular stratification instead of ECG [48]. A single measurement of BNP is pivotal for urgent decision making, although continuous monitoring may better define the prognosis [49]. Its shorter half-life (20 minutes) may limit its utility compared NT-pro-BNP. The latter is strongly associated with myocardial infarction, microsize one included, and fatal outcomes [50]. It may allow a quick risk stratification in emergency. Clinical practice guidelines on Heart Failure consider natriuretic peptides in class I of recommendation, level A of evidence for diagnosis and prognosis. Natriuretic peptide screening and guided therapy may prevent heart failure, reducing cardiovascular death and death by any cause [51]. A metanalysis reported that NT-pro-BNP concentration strongly predicts first-onset heart failure and risk of coronary heart disease and stroke [52]. Although the levels of high sensitive troponins seem to be related to the entity and rapidity of ischaemic process, their short half-lives may account for low positive predictive value, although higher their levels are, worst the outcomes. They seem to be more helpful for ruling out than ruling in acute myocardial infarction and no difference was reported in incidence of subsequent acute events or death from cardiovascular causes [53]. In severe acute stroke, a diffuse myocardial sufferance may be present, both for a chronic primary or secondary coronary hypoperfusion and stroke-related hyper-contracted state, responsible of coagulative myocytolisis and contraction band necrosis [54]. However, a misleading relative decline of high sensitive troponin levels may be observed because of reduced myocardial contractions and haemodilution in severe, end stage heart failure.
Our finding of significant fluctuations of NT-pro-BNP in class 0/I OND patients suggests that the burden may be wider than expected as well as the reversibility of physiological response versus a shift toward pathological dysfunctions. Any stressful event may interfere with physiological circulation, because of a prevalence of sympathic over parasympathic activity. NT-pro-BNP may be a warning marker of either an adaptive or maladaptive response. It is reported that the risk of cardiovascular disease in individuals with relatively low levels of NT-pro-BNP (55-124 pg/ml) was 1,9-fold higher than in those with the lowest levels (< 55 pg/ml) [55], independently of conventional risk factors [55,56]. Patients with elevated natriuretic peptides without heart failure may have an increased risk of developing symptomatic heart failure [57]. Indeed, NT-pro-BNP is the most sensible and useful parameter, reflecting atrial overload and myocardial spreading depression. These may stand for physiological preconditioning or evolve toward subtle, critical ischaemia. The shift toward irreversibility depends on ejection fraction, peripheral resistances, compliance, strictly linked and further exacerbated by cerebral anoxic-ischaemic damage. The compensatory response, reducing arterial pressure and inducing natriuresis and diuresis, may become pathological, triggering a cascade of events, culminating in the Virchow triad (reduced flow, endothelial damage, increased coagulability), with subsequent elevated risk of ischaemic sufferance. This may be signaled by the rise of T Tro, before the appearance of electrocardiographic signs. Longer the hypoperfusion, worst and irreversible the cardiological and cerebral outcomes are. In vitro, cerebral tissue is even more vulnerable than cardiac one [58]. In vivo, within few seconds of cerebral ischaemia, electroencephalogram activity ceases, as an energy sparing response [59,60]. The impairment of respiratory chain, anoxic depolarization and spreading depression extend from the ischemic core to the ischaemic penumbra; no reflow phenomenon and/or haemorrhagic infarction may occur, especially in decompensated heart failure. Either in acute or in chronic cerebrovascular conditions, a cranio- caudal cascade may contribute to "neurogenic stunned myocardium", which worsen cardiac and cerebral perfusion. Further studies are needed on the relation between cardiac biomarkers and entity of cerebral damage. Our results highlight that even late may still be brain in all AS, class III/IV, C/D NYHA, ACA included. However, poor collaterals, decreased perfusion, increased diffusion, retrograde venous leakage may account for futile recanalization and increase the risk of haemorrhagic complications.
Moreover, In AS in the context of CCVD, the concomitant presence of chronic myocardial ischaemic sufferance further increases the mismatch between diffusion and perfusion with subsequent risk of cerebral ischaemic core enlargement, oedema formation, mass effect on brainstem, diffuse anoxic ischaemic encephalopathy. Although some authors observed an increased risk of cardiac arrhythmia and death in AS with insular involvement [61], we did not find differences in cardiac biomarkers between insular and other AS. However, we do not exclude that an early and persistent rise of cardiac biomarkers in AS in cortical-subcortical encephalic regions, as insula, diencephalon and brainstem, may predict a faster necrotic evolution and account for malignant dysautonomia. Moreover, the higher risk of stroke in patients suffering from sleep apnoea [62] suggests that transient or persistent lacunar, ischaemic sufference may predispose to more severe ischaemic events, hinder the recovery and worsen the outcomes in vertebro-basilar AS, when respiratory centers in brainstem are early involved. Indeed, an association of sleep apnoea with clinically silent microvascular brain tissue changes in general population and in AS and between sleep disordered breathing and brainstem infarction are reported 1249 [63-65].
Conclusion
NT-pro-BNP and hs T Tro are pivotal for a prompt assessment of circulatory condition for the most appropriate treatment, directed to widen the therapeutic window and limit the costs related to invalidating outcomes. While the former is a sign of functional alteration, the latter represents already a structural damage. Although serum cardiac biomarkers may reflect more the chronicity rather than the acuity of ischaemic sufferance, delta criterion helps in differential diagnosis between acute versus chronic condition. Since apparently physiological conditions may evolve to subtle or overt pathological conditions, parallel haematological and urinary parameters, together with more sophisticated electrophysiological and neuroradiological techniques, may help in defining the burden and lesional load and establish the criteria for rational procedures. Further studies are needed for better defining the therapeutical window in AS patients suffering from heart failure, decision making concerning the strategy of "scoop and run" to mechanical thrombectomy or "stay and play" with plasminogen activators and/or other pharmacological agents 157 TOT 3270.
Acknowledgements
Declarations of conflict of interests: None.
We thank the colleagues of the Radiological Service of Criscuoli Hospital, S. Angelo dei Lombardi (AV).
References
- Christenson E, Christenson RH (2013) The role of cardiac biomarkers in the diagnosis and management of patients presenting with suspected acute coronary syndrome. Ann Lab Med 33: 309-318.
- McEvoy JW, Lazo M, Chen Y, Shen L, Nambi V, et al. (2015) Patterns and determinants of temporal change in high-sensitivity cardiac troponin-T: The atherosclerosis risk in communities cohort study. Int J Cardiol 187: 651-657.
- McEvoy JW, Chen Y, Ndumele CE, Solomon SD, Nambi V, et al. (2016) Six-year change in high-sensitivity cardiac troponin T and risk of subsequent coronary heart failure and death. JAMA Cardiol 1: 519-528.
- Jannuzzi Jr JL (2016) Biomarkers to predict risk in apparently well populations. JAMA Cardiol 1: 528-529.
- Faiz KW, Thommessen B, Einvik G, Haugar Brekle P, Omland T, et al. (2014) Determinants of high sensitivity cardiac troponin T elevation in acute ischemic stroke. BMC Neurol 14: 96.
- Faiz KW, Thommessen B, Einvik G, Omland T, Ronning OM (2014) Prognostic value of high-sensitivity cardiac troponin t in acute ischemic stroke. J Stroke Cerebrovasc Dis 23: 241-248.
- Furtner M, Ploner TF, Hammerer-Lercher AF, Pechlaner RF, Mair J (2012) The high-sensitivity cardiac troponin T assay is superior to its previous assay generation for prediction of 90-day clinical outcome in ischemic stroke. Clin Chem Lab Med 50: 2027-2029.
- James P, Ellis CJ, Withlock RML, McNeil AR, Henley J, et al. (2000) Relation between troponin T concentration and mortality in patients presenting with an acute stroke: observational study. BMJ 320: 1502-1504.
- Jensen JK, Kristensen SR, Bak S, Atar D, Hoilund-Carlsen PF, et al. (2007) Frequency and significance of troponin T elevation in acute ischemic stroke. Am J Cardiol 99: 108-112.
- Song HS, Back JH, Jin DK, Chung PW, Moon HS, et al. (2008) Cardiac troponin T elevation after stroke: Relationships between elevated serum troponin T, stroke location and prognosis. J Clin Neurol 4: 75-83.
- Stahrenberg R, Niehaus CF, Edelmann F, Mende M, Wohlfahrt J, et al. (2013) High-sensitivity troponin assay improve predicition of cardiovascular risk in patients with cerebral ischaemia. J Neurol Neurosurg Psy 84: 479-487.
- Apple FS, Jesse RL, Newby LK, Wu AHB, Christenson RH, et al. (2007) National academy of clinical biochemistry and IFCC committee for standardization of markers of cardiac damage laboratory medicine practice guidelines: Analytical issues for biochemical markers of acute coronary syndromes. Circulation 115: 352-355.
- Morrow DA, Bonaca MP (2013) Real-world application of "delta" troponin: diagnostic and prognostic implications. J Am Coll Card 62: 1239-1241.
- Redfield MM, Rodeheffer RJ, Jacobsen SJ, Mahoney DW, Bailey KR, et al. (2002) Plasma brain natriuretic peptide concentration: Impact of age and gender. J Am Coll Cardiol 40: 976-982.
- Wang TJ, Larson MG, Levy D, Leip EP, Benjamin EJ, et al. (2002) Impact of age and sex on plasma natriuretic peptide levels in healthy adults. Am J Cardiol 90: 254-258.
- McDonagh TA, Robb SD, Murdoch DR, Morton JJ, Ford I, et al. (1998) Biochemical detection of left ventricular systolic dysfunction. Lancet 351: 9-13.
- Smith H, Pickering RM, Struthers A, Simpson I, Mant D (2000) Biochemical diagnosis of ventricular dysfunction in elderly patients in general practice: Observational study. BMJ 320: 906-908.
- Davis M, Espiner E, Richards G, Billings J, Town I, et al. (1994) Plasma natriuretic peptide in assessment of acute dyspnoea. Lancet 343: 440-444.
- Dao Q, Krishnaswamy P, Kazanegra R, Harrison A, Aminovin R, Lenert L, et al. (2001) Utility of B-type natriuretic peptide in the diagnosis of congestive heart failure in an urgent-care setting. J Am Coll Card 37: 379-385.
- Cacciapuoti F (2010) Natriuretic peptide system and cardiovascular disease. Hearth Views 11: 10-15.
- Shahabi V, Moazenzadeh M, Azimzadeh BS, Nasri H, Afshar RM, et al. (2011) Relationship between serum N-terminal Pro Brain Natriuretic Peptide (NT-Pro BNP) level and the severity of coronary artery involvements. J Res Med Sci 16: 143-148.
- Logeart D, Thabut G, Jourdain P, Chavelas C, Beyne P, et al. (2004) Predischarge B-type natriuretic peptide assay for identifying patients at high risk of readmission after decompensated heart failure. J Am Coll Cardiol 43: 635-641.
- Salah K, Kok WE, Eurlings LW, Bettencourt P, Pimenta JM, et al. (2014) A novel discharge risk model for patients hospitalized for acute decompensated heart failure incorporating N-terminal pro-B-type natriuretic peptide levels: a European collaboration on Acute decompeNsated Heart Failure: ELAN- HF Score. Heart 100: 115-125.
- Rhee CK, Lim SY, Koh SO, Choi WI, Lee YJ, et al. (2014) Usefulness of N-terminal pro-B-type natriuretic peptide in patients admitted to the intensive care unit: a multicenter prospective observational study. BMC Anesthesiol 14: 16.
- Folsom AR, Nimbi V, Bell EJ, Oluleye OW, Gottesman RF, et al. (2013) Troponin T, N-terminal pro-B-type natriuretic peptide, and incidence of stroke: the atherosclerosis risk in communities' study. Stroke 44: 961-967.
- Pikula A, Beiser AS, De Carli C, Himal JJ, Debette S, et al. (2012) Multiple biomarkers and risk of clinical and subclinical vascular brain injury: The Framington offspring study. Circulation 125: 2100-2107.
- Rutten JH, Mattace-Raso FU, Steyerberg EW, Lindemans J, Hofman A, et al. (2010) Amino-terminal pro-B type natriuretic peptide improves cardiovascular and cerebrovascular risk prediction in the population: The Rotterdam study. Hypertension 55: 785-791.
- Takahashi T, Nakamura M, Onoda T, Ohsawa M, Tanno K, et al. (2009) Predictive value of plasma B type natriuretic peptide for ischemic stroke: a community-based longitudinal study. Atherosclerosis 207: 298-303.
- Wang TJ, Larson MG, Benjamin EJ, Leip EP, Omlamd T, et al. (2004) Plasma natriuretic peptide levels and the risk of cardiovascular events and death. N Engl J Med 350: 655-663.
- Campbell DJ, Woodward M, Chalmers JP, Colman SA, Jenkins AJ, et al. (2006) Soluble vascular cell adhesion molecule 1 and N-terminal pro-B-type natriuretic peptide in predicting ischemic stroke with cerebrovascular disease. Arch Neurol 63: 60-65.
- Kim SH, Lee JY, Park SH, Jang HC, Lim EJ, et al. (2013) Plasma B-type natriuretic peptide level in patients with acute cerebral infaction according to infarction subtype and infarction volume. Int J Med Sci 10: 103-109.
- Lynch JR, Blessing R, White WD, Grocott HP, Newman MF, et al. (2004) Novel diagnostic test for acute stroke. Stroke 35: 57-63.
- Makikallio AM, Makikallio TH, Korpelainen JT, Vuolteenaho O, Tapanainen JM, et al. (2005) Natriuretic peptides and mortality after stroke. Stroke 36: 1016-1020.
- Nakagawa K, Yamaguchi T, Seida M, Yamada S, Imae S, et al. (2005) Plasma concentration of brain natriuretic peptide in patients with acute ischemic stroke. Cerebrovasc Dis 19: 157-164.
- Nigro N, Wildi K, Mueller C, Schuetz P, Mueller B, et al. (2014) BNP is an independent prognostic marker for overall mortality inpatients with ischemic stroke or TIA and may improve the diagnostic accuracy to identify cardioembolic aetiology. PLoS One 9: e102704.
- Rost NS, Biffi A, Cloonan L, Chorba J, Kelly P, et al. (2012) Brain natriuretic peptide predicts functional outcome in ischemic stroke. Stroke 43: 441-445.
- Shibazaki K, Kimura K, Okada Y, Iguchi Y, Uemura J, et al. (2009) Plasma brain natriuretic peptide as an independent predictor of in hospital mortality after acute ischemic stroke. Intern Med 48: 1601-1606.
- Yukiiri K, Hosomi N, Naya T, Takahashi T, Ohkita H, et al. (2008) Plasma brain natriuretic peptide as a surrogate marker for cardioembolic stroke. BMC Neurol 8: 45.
- Daniels LB, Laughlin GA, Clopton P, Maisel AS, Barrett-Connor E (2008) Minimally elevated cardiac Troponin T and elevated N-Terminal Pro-B-Type Natriuretic Peptide predict mortality in older adults: Results from the Rancho Bernardo study. J Am Coll Cardiol 52: 450-459.
- de Lemos JA, Drazner MH, Omland T, Ayers CR, Khera A, et al. (2010) Association of troponin T detected with a highly sensitive assay and cardiac structure and mortality risk in the general population. JAMA 304: 2503-2512.
- Oluleye OW, Folsom AR, Nambi V, Lutsey PL, Ballantyne CM, et al. (2013) Troponin T, B type natriuretic peptide, C-reactive protein and cause specific mortality. Ann Epidemiol 23: 66-73.
- Thygesen K, Alpert JS, White HD (2007) Universal Definition of Myocardial Infarction. Circulation 116: 2634-2653.
- Victor RG, Haley RW, Willett DL, Peshock RM, Vaeth PC, et al. (2004) The Dallas heart study: A population-based probability sample for the multidisciplinary study of ethnic differences in cardiovascular health. Am J Cardiol 93: 1473-1480.
- Omland T, de Lemos JA, Sabatine MS, Christophi CA, Rice MM, et al. (2009) Prevention of events with angiotensin converting enzyme inhibition (PEACE) trial investigators. A sensitive cardiac troponin T assay in stable coronary artery disease. N Engl J Med 361: 2538-2547.
- Melki D, Lind S, Agewall S, Jernberg T (2012) Prognostic value of combining high sensitive troponin T and N terminal pro B type natriuretic peptide in chest pain patients with no persistent ST elevation. Clin Chim Acta 413: 933-937.
- Mathewkutty S, Sethi SS, Aneja A, Shah K, Iyengar RL, et al. (2013) Biomarkers after risk stratification in acute chest pain (from the BRIC Study). Am J Cardiol 111: 493-498.
- Maalouf R, Bailey S (2016) A review on B-type natriuretic peptide monitoring: Assays and biosensors. Heart Fail Rev 21: 567-578.
- Paget V, Legedz L, Gaudebout N, Girerd N, Bricca G, et al. (2011) N-terminal- pro-brain natriuretic peptide: A powerful predictor of mortality in hypertension. Hypertension 57: 702-709.
- Dhaliwal AS, Denswal A, Pritchett A, Aguilar D, Kar B, et al. (2009) Reduction in BNP levels with treatment of decompensated heart failure and future clinical events. J Card Fail 15: 293-299.
- Sterling MR, Durant RW, Bryan J, Levitan EB, Brown TM, et al. (2018) N-terminal pro-B-type natriuretic peptide and microsize myocardial infarction risk in the reasons for geographic and racial differences in stroke study. BMC Cardiovascular Disorders 18: 66.
- Yancy CW, Jessup M, Biykem Bozkurt, Javed Butler, Donald E CaseyJr, et al. (2017) 2017 ACC/AHA/HFSA focused update of the 2013 ACCF/AHA guideline for the management of heart failure: A report of the American college of cardiology/American heart association task force on clinical practice guidelines and the heart failure society of America circulation. 2017;136: e137-e161.
- Natriuretic Peptides Studies Collaboration, Willeit P, Kaptoge S, Welsh P, Butterworth AS, et al. (2016) Natriuretic peptides and integrated risk assessment for cardiovascular disease: an individual-partecipant-data meta-analysis. Lancet Diabetes Endocrinol 4: 840-849.
- Shah ASV, Anand A, Strachan FE, Ferry AV, Lee KK, et al. (2018) High-sensitivity troponin in the evaluation of patients with suspected acute coronary syndrome: A stepped-wedge, cluster-randomised controlled trial. Lancet 392: 919-928.
- Samuels MA (2007) The brain-heart connection. Circulation 116: 77-84.
- Doi Y, Ninomiya T, Hata J, Hirakawa Y, Mukaj N, et al. (2011) N- terminal pro-brain natriuretic peptide and risk of cardiovascular events in a Japanese community: the Hisayama Study. Arteriosclerosis, Thromb Vasc Biol 31: 2997-3003.
- Olsen MH, Kristian W, Nielsen Olav W, Hall C, Wergeland R, et al. (2006) N-terminal pro-brain natriuretic peptide predicted cardiovascular events stronger than high sensitivity C-reactive protein in hypertension: a LIFE substudy. J Hypertension 24: 1531-1539.
- Ledwidge M, Gallagher J, Conlon C, Tallon E, O'Connell E, et al. (2013) Natriuretic peptide-based screening and collaborative care for heart failure: the STOP-HF randomized trial. JAMA 310: 66-74.
- Lee JM, Grabb MC, Zipfel GJ, Choi DW (2000) Brain tissue responses to ischaemia. J Clin Invest 106: 723-730.
- Chen J, Simon R (1997) Ischemic tolerance in the brain. Neurology 48: 306-311.
- Murry CE, Jennings RB, Reimer KA (1986) Preconditioning with ischaemia: A delay of lethal cell injury in ischaemic myocardium. Circulation 74: 1124-1136.
- Abboud H, Berroir S, Labreuche J, Orjuela K, Amarenco P, et al. (2006) Insular involvement in brain infarction increases risk for cardiac arrhythmia and death. Ann Neurol 59: 691-699.
- Culebras A (2000) Sleep apnea and stroke. Rev Neurol Dis 2: 13-19.
- Kim H, Yun CH, Thomas RJ, Lee SH, Seo SH, et al. (2013) Obstructive sleep apnea as a risk factor for cerebral white matter change in a middle-aged and older general population. Sleep 36: 709-715.
- Kepplinger J (2014) Association of sleep apnea with clinically silent microvascular brain tissue changes in acute cerebral ischemia. J Neurol 261: 343-349.
- Brown DL, McDermott M, Mowla A, De Lott L, Morggenstern LB, et al. (2014) Brainstem infarction and sleep-disordered breathing in the BASIC sleep apnea study. Sleep Med 15: 887-891.
Keywords
INDEXING
PARTNERS









Table 1: Patients classification bases on their conditions. View Table 1