-
Address:
17888 67th Court North
Loxahatchee, FL
-
Mail us:
contact@wrightacademia.org
- submit manuscript
Table of content
Research Article |
Open Access |
Volume 1 | Issue 1 |
The Triage ICH Model for Predicting Prognosis of Patients with Non Traumatic Supratentorial Intracerebral Hemorrhage Admitted in Non ICU Setting: A Real World Brief Report
Luca Masotti1, Elisa Grifoni1, Alessandro Dei1, Rosario Spina2, Italo Calamai2, Giulia Guazzini1, Eleonora Cosentino1, Ester Latini1, Antonio Giordano1, Francesca Dainelli1, Francesca Maggi1, Chiara Giulietti1, Mario Romagnoli1, Stefano Cinotti1, Elena Schipani1, Giuseppe Salvatore Murgida1, Stefania Di Martino1, Andrea Cozzi1, Eleonora Sisti1, Adele Carli Ballola1, Debora Dacomo1, Alessandra Pratesi1, Ira Signorini1, Moira Bonfanti3, Elisabetta Daini3, Maria Letizia Bartolozzi4, Mariella Baldini4, Antonio Segneri5, Sandro Santini5, Cristina Moncini6, Elisa Migliorini6, Gaia Conti6, Gessica Tondini6, Stefania Moretti6, Leonardo Pellicciari6, Anna Bruni6, Nico Rosi7, Paola Bartalucci7 and Simone Vanni7
1Internal Medicine, San Giuseppe Hospital, Empoli, Florence, Italy
2Intensive Care Unit, San Giuseppe Hospital, Empoli, Florence, Italy
3Bed Manager, San Giuseppe Hospital, Empoli, Florence, Italy
4Neurology, San Giuseppe Hospital, Empoli, Florence, Italy
5Radiology, San Giuseppe Hospital, Empoli, Florence, Italy
6Physical Medicine and Rehabilitation, Empoli, Florence, Italy
7Emergency Department, San Giuseppe Hospital, Empoli, Florence, Italy
*Corresponding author: Luca Masotti, Head, Internal Medicine II, San Giuseppe Hospital, Viale Boccaccio 20, 50053, Empoli, Florence, Italy, E-mail: luca.masotti@tin.it
Citation: Masotti L, Grifoni E, Dei A, Spina R, Calamai I, et al. (2019) The Triage ICH Model for Predicting Prognosis of Patients with Non Traumatic Supratentorial Intracerebral Hemorrhage Admitted in Non ICU Setting: A Real World Brief Report. Academia J Stroke. 1:001.
Copyright © Masotti L, et al.
Received: |
Accepted: |
Published: |
Background and aim
Appropriate setting of care based on mortality risk is of utmost importance for reducing adverse outcome in patients with non-traumatic intracerebral hemorrhage (ICH). The Triage ICH model, which includes as variables Glasgow Coma Scale score < 13, ICH volume ≥ 30 mL and intraventricular bleeding, has been proposed as effective and safe tool for identifying patients with supratentorial ICH requiring Intensive Care Unit (ICU) admission.
The aim of the present study was to analyze the predictive value of Triage ICH model in patients admitted in non ICU setting.
Materials and methods
We retrospectively analyzed clinical and neuro-radiological data of patients admitted for ICH in a dedicated non ICU stroke area. In-hospital mortality and composite endpoint death and/or severe disability at discharge according to the Triage ICH model scoring were the study outcomes.
Results
One hundred and seventy-five patients (46.8% females) with mean age ± SD 79.2 ± 10.8 years were the study population. Fifty-nine patients (33.7%) had ICH volume ≥ 30 mL, forty-seven (26.8%) had GCS score < 13 and sixty-seven (38.2%) had IVH. Eighty-seven patients (49.7%) were classified to have Triage ICH model score 0, whereas ninety-three (50.3%) had at least one of the three variables included in the model. Overall, fifty-two patients (29.7%) developed an early neurological worsening (ENW) and fifty-six (32%) died during hospitalization. In-hospital mortality and composite endpoint death and/or mRS ≥ 4 increased from 6.8% and 39% respectively in patients with Triage ICH model score 0 (none of the three variables present) to 93.7% and 100% respectively in patients with Triage ICH model score 3 (all the three variables present). The AUC was 0.89 for in-hospital mortality and 0.98 for the composite endpoint death and/or severe disability. Patients with Triage ICH model score 0 had significantly lower percentage of non lobar ICH, hematoma enlargement, ENW and modified Rankin Scale score ≥ 4 compared with patients with at least one of the three variables included in the model.
Conclusion
The Triage ICH model has high prognostic predictive value in patients with supratentorial ICH admitted in non ICU setting. The contemporary absence of ICH volume ≥ 30 mL, GCS score < 13 and IVH seems to identify a subgroup of patients with low mortality and severe disability risks who could be safely managed in non ICU setting.
Introduction
Identifying the appropriate setting of care based on risk mortality is of utmost importance to improve the prognosis of patients with non traumatic intracerebral hemorrhage (ICH). International guidelines recommend that patients suffering for ICH be admitted to either Intensive Care Unit (ICU) or dedicated Stroke Unit (SU) with acute neuroscience expertise [1]. However, in clinical practice many factors could influence the choice to admit patients in ICU or non ICU settings such as patients' co-morbidity, physicians' attitude to ICH management and local organization. Recently, Klaas JP, et al. derived and validated the Triage ICH model (TICH model) as effective and safe model to predict ICU admission in patients with supratentorial ICH [2]. The model utilizes three variables: Glasgow Coma Scale (GCS) score < 13, ICH volume ≥ 30 mL and intraventricular bleeding (IVH) each of them weights for one point. The presence of at least one of the three variables identifies patients requiring ICU admission, whereas the absence of all the three variables identifies patients not requiring ICU. In the validation cohort, the Triage ICH model predicted ICU admission with a sensitivity of 97.8% (AUC 0.88) [2]. In their article, the Authors concluded that patients with none of the abovementioned factors upon initial presentation could be safely triaged to non-ICU level of care (Table 1) [2].
The aim the present study was to evaluate the predictive power of TICH model regard the in-hospital mortality and severe disability at discharge of supratentorial ICH patients triaged in Emergency Department for non-ICU level of care.
Materials and Methods
We retrospectively analyzed clinical and neuroradiological data of consecutive patients suffering for supratentorial non traumatic ICH and firstly triaged for non ICU admission in Emergency Department (ED). All patients were allocated in a dedicated Stroke Area of our Hospital. For all patients enrolled the severity of neurological impairment at ED admission evaluated by GCS, early neurological worsening (ENW) defined as GCS score fall ≥ 2 points within 48 hours from symptoms onset, ICH volume calculated by using the ABC/2 formula [3], IVH and its entity calculated by using Graeb score [4] were registered. Hematoma enlargement was evaluated on a second CT scan, and was defined as significant for an ICH volume increase of one third in comparison with basal brain CT scan, or for an increase of 6 mL when ICH volume was < 30 mL. Hemphill ICH score was calculated for each patient according to the study validation [1]. In-hospital mortality and composite endpoint death and/or severe disability at hospital discharge defined as a modified Rankin Scale (mRS) score ≥ 4 were the examined prognostic outcomes.
Statistical analysis
Continuous variables were reported as mean ± standard deviation (SD) when normally distributed, as median and interquartile range (IQR) when not normally distributed. ICH score was reported as median (IQR). Categorical variables were analyzed by using test and Fisher exact test, as appropriate. For evaluating the TICH model score prognostic ability the area under the curve (AUC) of Receiver Operating Characteristic (ROC) curve was calculated. All p-values of < 0.05 were considered statistically significant.
Results
One hundred and seventy-five patients (46.8% females) with mean age ± SD 79.2 ± 10.8 years were the study population. Characteristics of patients are summarized in Table 2. Fifty-nine patients (33.7%) had ICH volume ≥ 30 mL, forty-seven (26.8%) had GCS score < 13 and sixty-seven (38.2%) had IVH. Median Graeb score was 4.5 (IQR 2-8). Eighty-seven patients (49.7%) were classified to have TICH model score 0, whereas eighty-eight (50.3%) had at least one of the three variables included in the model. Overall, fifty-two patients (29.7%) developed an early neurological worsening (ENW) and fifty-six (32%) died during hospitalization. Nine patients (5.1%) with mean age 67 ± 15 years, four of them classified as TICH model 0 and five with TICH model ≥ 1, required ICU transfer. The main reason for ICU transfer was ENW which occurred in six of nine patients. In-hospital mortality increased from 6.8% in patients with TICH model score 0 (none of the three variables present) to 93.7% in patients with TICH model score 3 (all the three variables present). The composite outcome death and/or severe disability at discharge increased from 39% in patients with TICH model score 0 to 100% in patients with TICH model score ≥ 2) (Table 3 and Figure 1). The AUC for in-hospital mortality was 0.89 (R^2 = 0.9735), whereas the AUC for the composite outcome death and/or severe disability was 0.98 (R^2 = 0.8676). Patients with TICH model score 0 had significantly lower percentage of non lobar ICH (67.8% vs. 51.2%, p = 0.0311), hematoma enlargement (4.6% vs. 28%, p = 0.0001), ENW (8% vs. 51.1%, p = 0.0001) and modified Rankin Scale score ≥ 4 (39% vs. 97.7%, p = 0.0001) compared with patients with TICH model ≥ 1 (Table 4).
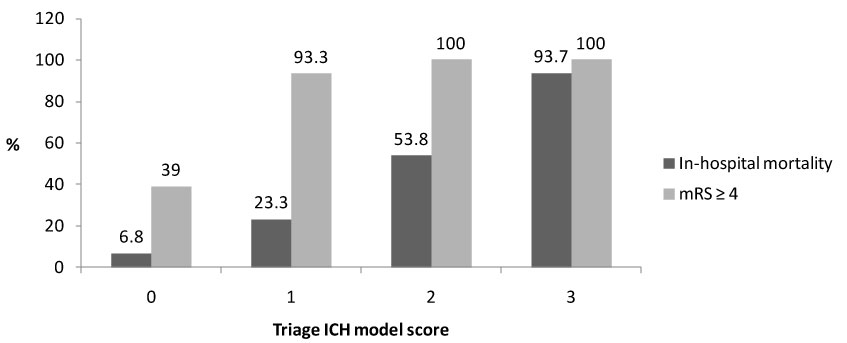
Figure 1: Prognostic outcomes according to Triage ICH model.
Table 2: Characteristics of study population. View Table 2
Table 3: Triage ICH Model, in-hospital mortality and severe disability at discharge. View Table 3
Table 4: Comparison between patients with TICH model 0 with patients with TICH model ≥ 1. View Table 4
Discussion
Non traumatic ICH remains a devastating disease due to high mortality risk and severe functional sequels. Thity-day and one year mortality are about 40% and 50% respectively; after one year from ICH, less than one third of survivors is totally independent [3]. In the latest years many efforts have been made to improve prognostic stratification. The main negative prognostic factors are Glasgow Coma Scale (GCS) score ≤ 9, ENW, ongoing anticoagulants and antiplatelets therapy, age ≥ 80 years, elevated systolic blood pressure (SBP), hyperglicemia, hyponatriemia, infratentorial ICH site, ICH volume > 30 mL, IVH, midline shift, hydrocephalus, spot sign on computed tomography (CT) angiography, and hematoma enlargement on second CT scan [1,4-6]. Some of these variables have been included in prognostic scores, such as the Hemphill ICH score, which estimates 30-day mortality [7]. Despite supratentorial ICH seems to have better outcomes compared with infratentorial ICH, the prognostic burden of supratentorial ICH is not negligible even if the ICH volume is less than 30 mL. In a previous study Behrouz R, et al. found among 375 cases of supratentorial ICH with volume < 30 mL ICH enlargement and ENW rates of 19.2% and 7.5%, respectively [8]. In-hospital mortality and/or thirty-day severe disability (mRS 4-5) occurred in 42.9% of patients. Age, admission GCS score, IVH and ENW were independent risk factors for poor outcome [8].
ICU represents the most appropriate setting of care for a great proportion of patients with non traumatic ICH. Moreover, neurological/neurosurgical ICU seem to guarantee better outcomes compared to general ICU [9]. However ICU admission could be not cost effective in patients with mildly or moderately severe ICH. Thus, the identification of the appropriate setting of care based on risk mortality is of the main importance in patients with ICH. Needing for intubation/mechanical ventilation and/or neurosurgical procedure, the presence of respiratory failure and/or two or more organ failures, significant clinical and/or neurological deterioration are recognized criteria for ICU admission worldwide. As abovementioned, in the study of Klass JP, et al. the presence of at least one of GCS score < 13, ICH volume ≥ 30 mL and IVH identified patients requiring ICU admission [2]. In the INTERACT II trial younger age, recruitment in China, prior ischemic/undetermined stroke, high systolic blood pressure, National Institute of Health Stroke Scale (NIHSS) score ≥ 15, ICH volume ≥ 15 mL, IVH, early neurological deterioration, intubation and surgery were found independent risk factors for ICU admission [10].
Patients with ICH seem to benefit at least as much as patients with ischemic stroke from SU. In a meta-analysis of thirteen trials recruiting a total of 3570 patients with ICH, deaths or dependency were not different in patients with ICH or ischemic stroke who were admitted in SU [11]. Therefore SU or dedicated Stroke areas or dedicated Stroke areas could be an alternative to ICU in a subgroup of patients suffering for ICH. The accurate patients' selection becomes a cornerstone of modern management of ICH. In a previous study Alkhachroum AM, et al. found in a cohort of 431 patients that the contemporary presence of supratentorial ICH, ICH volume < 20 mL, no evidence of IVH, no respiratory failure, GCS ≥ 12 and SBP < 200 mmHg identified ICH patients with low risk of adverse outcome (in-hospital mortality 1.41%) [12]. Fehnel CR, et al. compared patients with small supratentorial ICH (≤ 20 mL) admitted in ICU (41 patients) or in or in SU (63 patients) [13]. The Authors found a non significant lower rate of poor functional status (mRS ≥ 3) at discharge in patients admitted in SU compared with patients admitted in ICU (57% vs. 76%, p = 0.06), in-hospital mortality (13% vs. 20%, p = 0.35) and complications (19 vs. 27%, p = 0.35), whereas patients admitted in ICU had a longer median hospital stay (3 vs. 6 days, p < 0.001) [14]. In another study Hafeez S, et al. reported outcomes of twenty patients with mild presentation ICH admitted in SU instead of ICU [14]. In this study mild ICH was defined as combination of Hemphill ICH score ≤ 2, NIHSS score ≤ 15 and Graeb score ≤ 2 in patients with IVH. No patient was transferred to ICU and in-hospital mortality was 5% [14].
In the present study, we focused on outcomes of patients with supratentorial ICH firstly triaged for non ICU setting and admitted in a dedicated Stroke area. We tested the predictive value of the Triage ICH model on in-hospital mortality and composite endpoint death and/or severe disability at hospital discharge. Findings from the present study showed that the Triage ICH model has high predictive value in prognosticating in-hospital mortality and severe disability at discharge (AUC 0.89 and 0.98, respectively) and it could be an optimal tool aimed to select the appropriate setting of care for patients with supratentorial ICH. Of note, the contemporary absence of ICH volume ≥ 30 mL, GCS score < 13 and IVH seems to identify a subgroup of patients with low in-hospital mortality and severe disability risks who could be managed in non ICU setting. In our study, in fact, patients with TICH model score 0 had 8.3 fold reduced risk of in-hospital mortality and 2.5 fold reduced risk of severe disability at discharge compared with patients with TICH model score ≥ 1.
We recognize that our study has limitations such as the small sample size, the retrospective methodology and the single center location. However, it could add important information about ICH management.
Conclusion
Defining the appropriate setting of care is fundamental in patients with non traumatic ICH. Our real life report seems to confirm that the Triage ICH model could be an optimal tool for identifying a subgroup of patients at low risk of ENW and/or in-hospital mortality who could be managed safely in non ICU setting.
Aknowledgments
The Authors thank all physicians, nurses and physiotherapists of the Emergency Department, Radiology, Intensive Care Unit, Sub-Intensive Care Unit, Stroke Unit, Internal Medicine II wards and Rehabilitation Unit of San Giuseppe Hospital of Empoli (Florence), Italy, all physicians, nurses and physiotherapists of Physical Medicine and Rehabilitation Unit of San Miniato Hospital (Pisa), Italy and all physicians of Neurosurgery Unit and Neurointensive Care Unit of Careggi Hospital (Florence), Italy, for the contribute and collaboration in management of patients suffering for ICH.
References
- Hemphill JC, Greenberg SM, Anderson CS, Becker K, Bendok BR, et al. (2015) Guidelines for the management of spontaneous intracerebral hemorrhage: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 46: 2032-2060.
- Klaas JP, Braksick S, Mandrekar J, Sedova P, Bellolio MF, et al. (2017) Factors associated with the need for intensive care unit admission following supratentorial intracerebral hemorrhage: The Triage ICH Model. Neurocrit Care 27: 75-81.
- Broderick JP, Brott TG, Duldner JE, Tomsick T, Huster G (1993) Volume of intracerebral hemorrhage. A powerful and easy-to-use predictor of 30-day mortality. Stroke 24: 987-993.
- Graeb DA, Robertson WD, Lapointe JS, Nugent RA, Harrison PB (1982) Computed tomography diagnosis of intraventricular hemorrhage. Etiology and prognosis. Radiology 143: 91-96.
- Godoy DA, Piñero GR, Koller P, Masotti L, Di Napoli M (2015) Steps to consider in the approach andmanagement of critically ill patient with spontaneous intracerebral hemorrhage. World J Crit Care Med 4: 213-229.
- Pinho J, Costa AS, Araújo JM, Amorim JM, Ferreira C (2019) Intracerebral hemorrhage outcome: A comprehensive update. J Neurol Sci 398: 54-66.
- Hemphill JC, Bonovich DC, Besmertis L, Manley GT, Johnston SC (2001) The ICH score: A simple, reliable, grading scale for intracerebral hemorrhage. Stroke 32: 891-897.
- Behrouz R, Misra V, Godoy DA, Topel CH, Masotti L, et al. (2017) Clinical course and outcomes of small supratentorial intracerebral hematomas. J Stroke Cerebrovasc Dis 26: 1216-1221.
- Diringer MN, Edwards DF (2001) Admission to a neurological/neurosurgical intensive care unit is associated with reduced mortality rate after intracerebral hemorrhage. Crit Care Med 29: 635-640.
- Wartenberg KE, Wang X, Muñoz-Venturelli P, Rabinstein AA, Lavados PM, et al. (2017) Intensive Care Unit Admission for Patients in the INTERACT2 ICH blood pressure treatment trial: Characteristics, predictors, and outcomes. Neurocrit Care 26: 371-378.
- Langhorne P, Fearon P, Ronning OM, Kaste M, Palomaki H, et al. (2013) Stroke Unit care benefits patients with intracerebral hemorrhage: Systematic review and meta-analysis. Stroke 144: 3044-3049.
- Alkhachroum AM, Bentho O, Chari N, Kulhari A, Xiong W (2017) Neuroscience step-down unit admission criteria for patients with intracerebral hemorrhage. Clin Neurol Neurosurg 162: 12-15.
- Fehnel CR, Glerum KM, Wendell LC, Potter NS, Silver B, et al. (2018) Safety and costs of stroke unit admission for select acute intracerebral hemorrhage patients. Neurohospitalist 8: 12-17.
- Hafeez S, Behrouz R (2016) The safety and feasibility of admitting patients with intracerebral hemorrhage to the step-down Unit. J Intensive Care Med 31: 409-411.
INDEXING
PARTNERS









Table 1: The Triage ICH model. View Table 1